-
Analyzers
- Sulfur Analyzers
- Chlorine Analyzers
- Multi Element Analyzers
-
Parts and Supplies
Accessories PartsSupplies
- Service & Maintenance
- Optics & Sources
- Technologies
- Support
- About
Sulfur in Ethanol for Tier 3 Compliance
BACKGROUND
The use of biofuels has been growing over the past decade as the industry adds renewable sources to the commercial product mix. This trend has seen the rise of complete fuel products such as biodiesel, as well as bioethanol blend stocks for gasoline. Most automobiles in the U.S. are able to run on gasoline with a 10% mixture of fuel ethanol (E10). Since 2014, over half of the new vehicles produced by Chrysler, Ford, and General Motors can use E85, which is a mixture of 15% gasoline and 85% fuel ethanol. The growth in U.S. production of Fuel Ethanol from roughly 800k barrels per day in 2013 to over 1M barrels per day 1 in 2018 highlights the strong demand for these products. Since Jan 1st, 2017, ethanol that is blended with gasoline must meet the 10 parts per million (ppm) standard for sulfur content on a per gallon basis.
Ethanol produced through the fermentation of sugars from plants such as corn or sugar cane, sometimes referred to as bioethanol, is not a significant source of sulfur content in fuels. To restrict human consumption, ethanol is typically denatured with gasoline from various sources (including as a byproduct of natural gas production), which can potentially introduce sulfur content into the finished product. Denaturants cannot exceed 3% by volume of the fuel ethanol and can be sourced from a certified supplier or be subject to testing requirements. Certified ethanol denaturant must contain less than 330 ppm sulfur. To ensure compliance with Tier 3 regulations, fuel ethanol is tested to confirm that the sulfur content meets the 10 ppm standard for downstream-blended oxygenate.
CHALLENGE
Measuring sulfur content in fuel ethanol shares many of the same challenges as other biofuels. The oxygen content in ethanol can create an interference that masks the presence of sulfur. This makes it more difficult to quantify for sulfur and requires specific countermeasures to ensure accurate reporting. The US EPA provides specific guidance, stating:
We believe that ASTM D2622, the designated method for testing for sulfur content of gasoline, will be useable for this testing purpose, as long as the calibration of the instrument is performed with an ethanol blend that is representative of the samples that are expected to be tested.
In this paper we will introduce a calibration process for Sindie +Cl that uses matrix matching. Using a matrix matched calibration, we will demonstrate that a check sample, an ethanol Certified Reference Standard with 5 ppm sulfur, is measured with excellent agreement to the certified value.
EXPERIMENT
As with other samples that require oxygen correction, the first step is to measure calibration standards with sample matrices consistent with the samples being measured. We tested seven ethanol calibration standards with sulfur concentrations of 0, 0, 5, 15, 50, 250 and 500 ppm using Sindie +Cl in ASTM D7039 mode. The individual samples were vented and measured for 300 seconds each in low range.

RESULTS
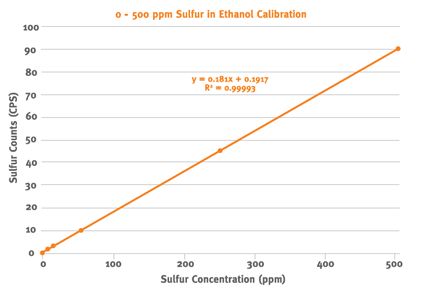
Figure 1 shows the calibration measurements along with the fitted calibration curve. The correlation coefficient (R2) of the calibration curve indicates an excellent fit to the measured values. The performance of the instrument was verified using 10 separate check samples with a certified sulfur concentration of 5 ppm. Each sample was vented and measured for 300s. Table 1 shows the measured concentrations of the check samples and demonstrates that there is no bias compared to the 5 ppm accepted reference value. The relative standard deviation for the 10 measurements is 8%.
| Table 1: 5 ppm Sulfur in Ethanol Check Sample | |
|---|---|
| Sample # | Concentration (ppm) |
| 1 | 5.1 |
| 2 | 5.3 |
| 3 | 5.0 |
| 4 | 6.0 |
| 5 | 4.4 |
| 6 | 4.9 |
| 7 | 4.9 |
| 8 | 4.7 |
| 9 | 4.9 |
| 10 | 5.1 |
| Average | 5.0 |
| Std Dev | 0.4 |
| RSD | 8% |
Figure 1: Sulfur in Ethanol Calibration
CONCLUSION
As with gasoline, blending ethanol is subject to US EPA Tier 3 regulations, including limits on the concentration of sulfur. While bioethanol derived from plants typically contains sulfur concentrations well below the regulatory limit, the denaturing process can potentially produce fuel ethanol which is non-compliant. By setting up a matrix matched calibration, batch testing with Sindie +Cl can provide a quick, easy-to-use method to check sulfur concentrations and ensure compliance.
Sindie +Cl utilizes a technique known as Monochromatic Wavelength Dispersive X-ray Fluorescence (MWDXRF) – delivering exceptional precision. It delivers trace analysis for both sulfur and chlorine with one push of a button and zero hassle. Samples are measured directly, which means it can analyze even the heaviest of hydrocarbons like crude oil or coker residuals, without the hassle of boats, injectors, furnaces, or changing detectors. Sindie +Cl provides refi neries, terminals, and third-party testing labs with an easy to use, highly precise elemental analyzer. METHOD COMPLIANCE: ASTM D2622, D7039, D4929, D7536; SH/T 0842.
Author: Joseph Iaia - Product Manager, Petroleum


